Case Study / Biopharmaceuticals
Launching AUCATZYL
Across Four Digital Properties
Autolus Therapeutics was preparing to launch AUCATZYL — a first-in-class CAR T cell therapy for relapsed or refractory B-cell acute lymphoblastic leukemia. As agency of record, Inizio Evoke was responsible for the entire digital launch ecosystem. I led UX strategy and design direction across four interconnected properties: the AUCATZYL HCP site, the AUCATZYL patient site, the Autolus Assist support platform, and a campaign landing page — four properties, two radically different audiences, one brand, and zero margin for error in a regulated pharmaceutical environment.
launched as a unified ecosystem
professionals and patients/caregivers
with audience-specific visual identities
Strategic Challenge
CAR T cell therapy sits at one of the sharpest intersections in healthcare communications: a treatment so scientifically advanced that it requires genuine clinical fluency to explain, serving patients who are often in the most frightening moment of their lives. Three compounding challenges defined the engagement.
"Four properties, two audiences, one brand — and every content decision subject to medical, legal, and regulatory review. The design process was as much about stakeholder alignment as creative execution."
First, the content itself: CAR T cell mechanisms, treatment protocols, eligibility criteria, side effect profiles — genuinely complex, requiring constant calibration between scientific accuracy for clinicians and accessibility for patients. Second, two audiences with fundamentally different needs had to be served across properties that felt related but distinct. Third, a regulated pharmaceutical environment meant every information hierarchy choice carried compliance implications — making regulatory alignment a design constraint to be built in from the start, not a filter applied at the end.
Discovery & Strategy
I led a discovery phase grounded in stakeholder interviews with Autolus's medical, product, and marketing teams, combined with a competitive audit of CAR T therapy and adjacent biotech digital experiences. The goal was to build genuine scientific fluency in the therapy and identify where category norms were failing both audiences.
Method 01
Stakeholder Interviews & Scientific Immersion
Directed sessions with Autolus's medical, product, and marketing teams to build genuine fluency in CAR T cell therapy — understanding not just what AUCATZYL does but how oncologists evaluate and administer it, and what patients and caregivers actually need to know to make informed decisions. Scientific accuracy was a design constraint from day one.
Method 02
Competitive Audit
Audited CAR T therapy and adjacent oncology digital experiences to identify where the category set the bar and where it consistently failed. A clear gap emerged: sites built for HCPs were dense and navigation-heavy; sites built for patients were emotionally appropriate but undersupported on practical information. Each failure mode was directly relevant to what the AUCATZYL ecosystem needed to solve.
Four Properties, One System
Each property needed a distinct structural logic reflecting its primary audience's actual workflow. The IA work was where the four-property strategy became concrete — and where the design system's role as connective tissue became essential. I directed the development of a shared component library that maintained brand coherence across all four properties while allowing audience-specific visual identities on top.

Property 01
AUCATZYL HCP
Built for oncologists and treatment center staff evaluating AUCATZYL for eligible patients. Content hierarchy led with clinical data and a restrained, authoritative visual palette that signaled the professional register immediately.
- Efficacy outcomes and safety profile surfaced at entry
- Patient selection criteria and dosing protocols clearly delineated
- Downloadable resources immediately accessible
- Content density calibrated to clinicians comfortable with detail
Property 02
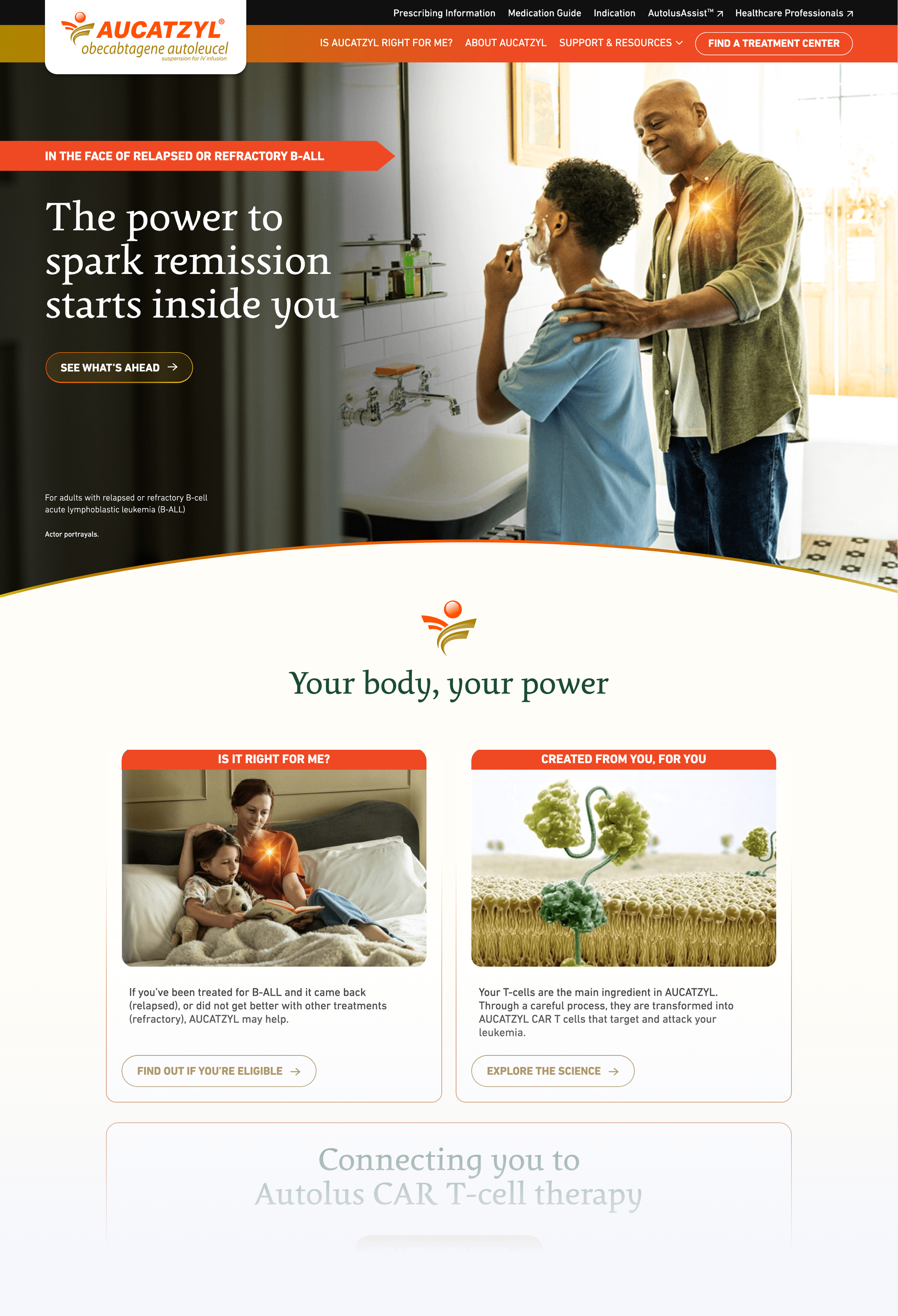
AUCATZYL Patient
Built for patients and caregivers navigating a treatment decision under significant emotional pressure. Content hierarchy followed the emotional arc of a patient's experience before addressing clinical mechanism — what to expect first, how it works second.
- Treatment process and side effect information sequenced progressively
- Warmer, more accessible visual treatment reflecting the patient register
- Questions to ask a doctor surfaced at key decision moments
- Language calibrated for non-specialist comprehension without sacrificing accuracy
Property 03
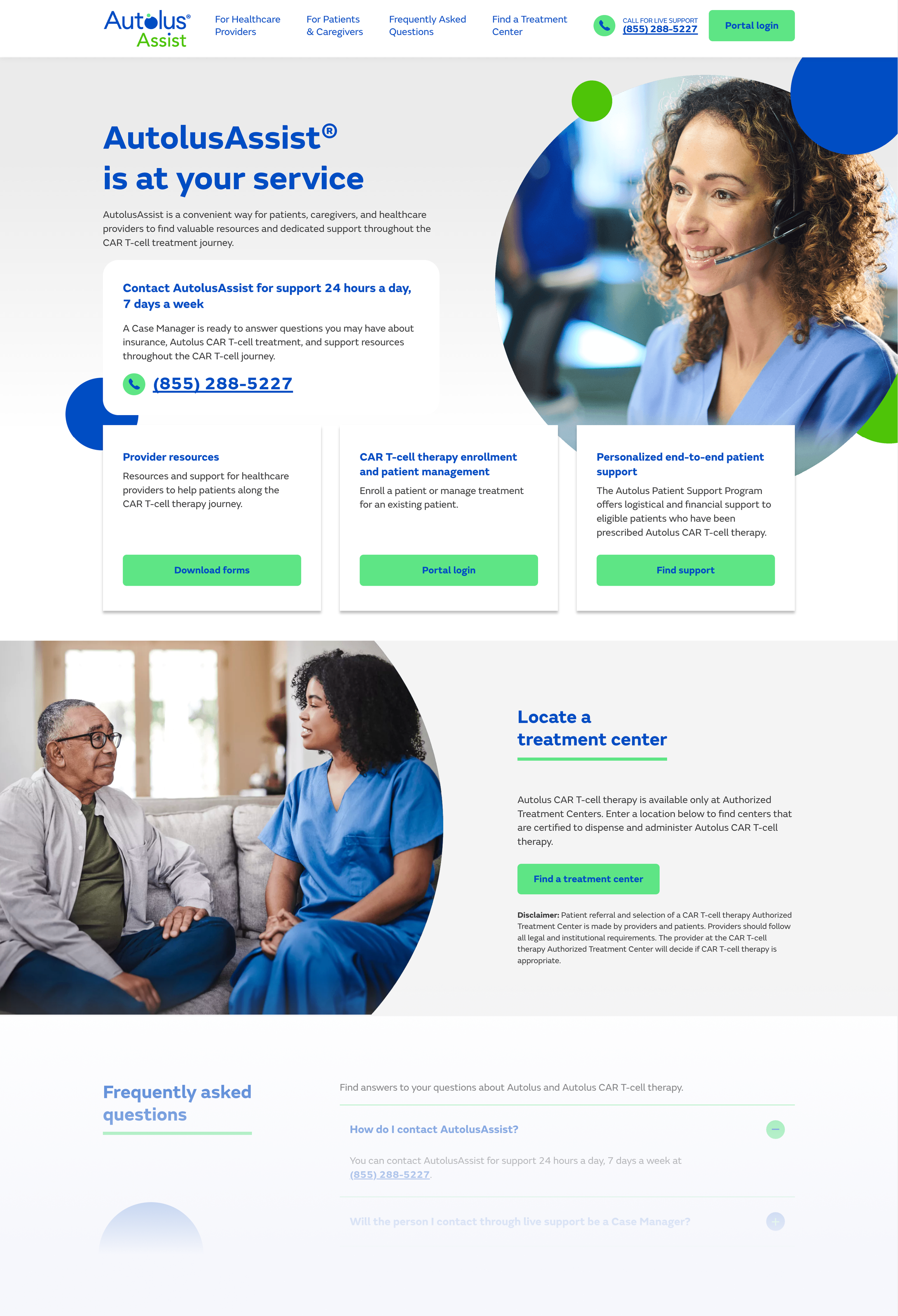
Autolus Assist
A support platform serving patients, caregivers, and HCPs navigating the practical logistics of CAR T therapy. Three distinct user types from a single entry point — requiring a clear audience-selection model that routed each user to the right resources without creating confusion.
- Audience-selection entry model routing each user type cleanly
- Interactive treatment site locator for authorized AUCATZYL centers
- Downloadable resources and dedicated support line access
- Visual treatment bridging clinical and patient registers simultaneously

Property 04
Autolus Campaign Page
A flagship brand expression showcasing Autolus's scientific leadership and the breakthrough nature of AUCATZYL — designed to work as both an introduction for new audiences and a credibility anchor for those already familiar with the therapy.
- Scientific leadership and breakthrough positioning at the forefront
- Accessible to new audiences without requiring prior CAR T knowledge
- Scroll-triggered animations explaining the mechanism of action
- Cohesive with the three core properties while standing alone as a brand statement
Design & Interactive Direction
With IA approved across all four properties, I directed visual design and interactive experience in close collaboration with Autolus's medical and regulatory review process — every design decision that touched content presentation developed with compliance implications in mind.
Visual Differentiation Within a Unified System
The HCP palette was deliberately restrained: clinical, precise, credibility-forward. Patient and Assist properties used warmer, more accessible color treatments. Both felt unmistakably Autolus. The differentiation was tonal — the underlying component system was shared across all four, ensuring brand coherence without visual uniformity.
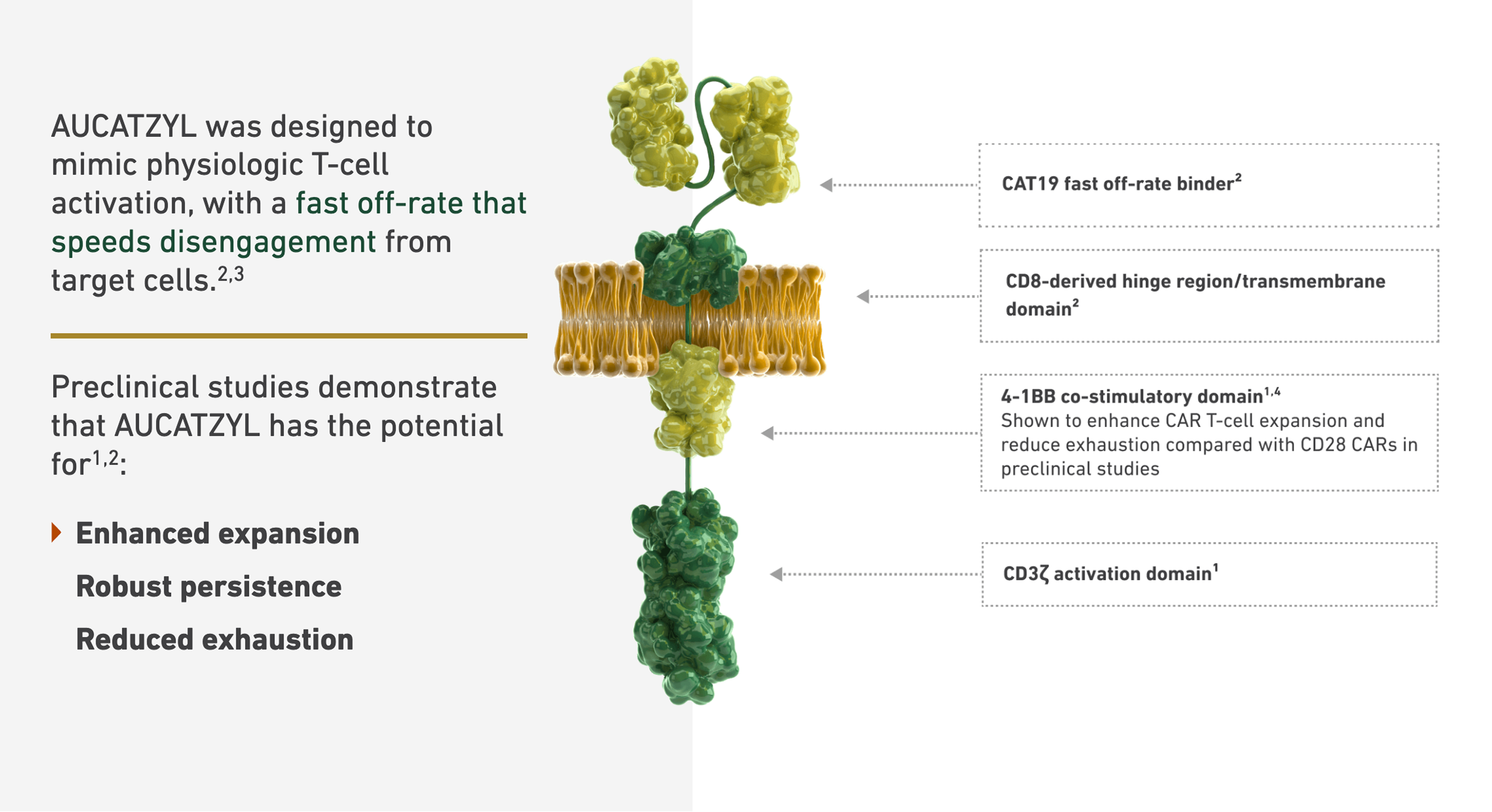
Mechanism of Action Animation
CAR T cell therapy is difficult to explain in static content. Scroll-triggered animations walked users through the mechanism of action in a way that was scientifically accurate and genuinely comprehensible to a non-specialist — the single most impactful design decision in the ecosystem for bridging the gap between clinical complexity and patient understanding.
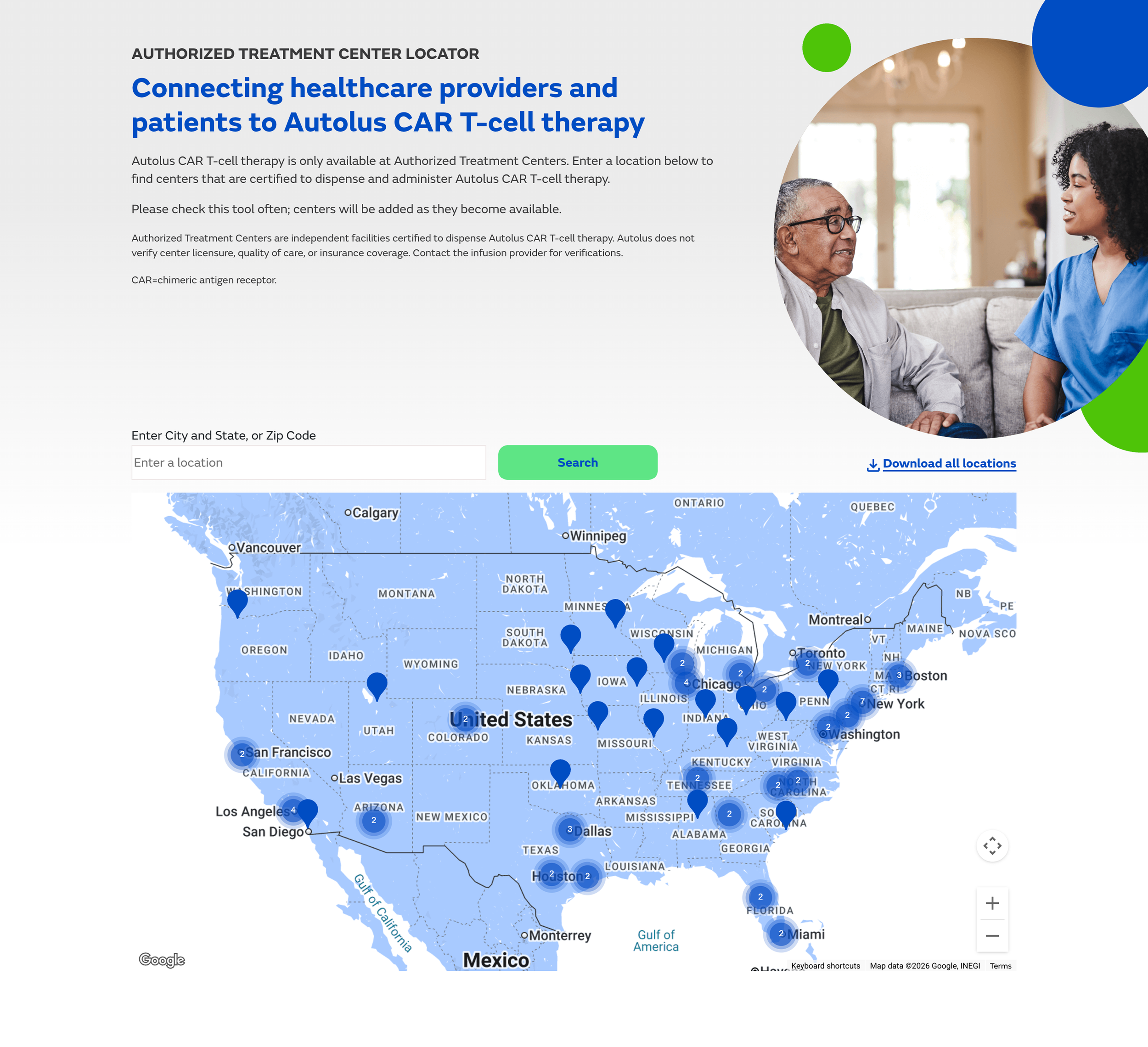
Interactive Treatment Site Locator
An interactive map surfacing authorized AUCATZYL treatment center locations gave both patients and clinicians immediate access to a practically critical piece of information. For patients navigating a serious diagnosis, finding a qualified treatment center is often the first urgent question — the locator answered it without requiring a search outside the platform.
Mobile-First Execution
The entire ecosystem was designed mobile-first — patients and caregivers access healthcare information on mobile, often in clinical settings or during high-stress moments at home. Every interaction, layout, and content density decision was made for a mobile context first, then scaled to desktop.
Regulatory-Integrated Design Process
Working within pharma regulatory constraints means understanding that IA choices, label proximity, safety information hierarchy, and content emphasis all carry compliance implications. I built those constraints into the design process from the first wireframe rather than treating MLR review as a post-design filter — reducing revision cycles and protecting launch timelines.
Scalable Design System
A component library — buttons, forms, icons, cards, interactive elements — shared across all four properties, with audience-specific color schemes applied on top. Gave Autolus the infrastructure to extend the platform to new indications or markets without rebuilding from scratch at each expansion.
Design Point 02 · Mechanism of Action
Design Point 03 · Interactive Treatment Site Locator
Design Point 01 · Visual Differentiation — Campaign Register

Design Point 01 · Clinical Data Presentation — HCP Site

Outcomes & Impact
The AUCATZYL digital ecosystem launched as a coherent, multi-property brand presence — giving Autolus the platform infrastructure to support one of oncology's most significant recent therapy launches.
Unified Digital Ecosystem
Four properties launched as a coherent brand experience — each purpose-built for its audience while operating from a single design system. A clinician moving between the HCP site and Autolus Assist encountered a consistent Autolus experience calibrated appropriately for each context.
Increased Patient Engagement
Patients and caregivers reported the information was more accessible and less overwhelming than prior digital touchpoints — with measurable increases in time on site validating that the content architecture was meeting users where they were rather than front-loading complexity.
Regulatory-Compliant by Design
All properties designed and executed with pharmaceutical regulatory constraints integrated into the process — not applied after the fact. The result was a faster, more efficient MLR review cycle and a launch-ready product that hadn't been compromised by late-stage compliance revisions.
Scalable Platform Foundation
A shared component library giving Autolus the infrastructure to extend the platform — new indications, additional markets, future therapy launches — without rebuilding from scratch. An investment in long-term platform sustainability, not just the AUCATZYL launch.
